AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Anti mog myelitis9/16/2023  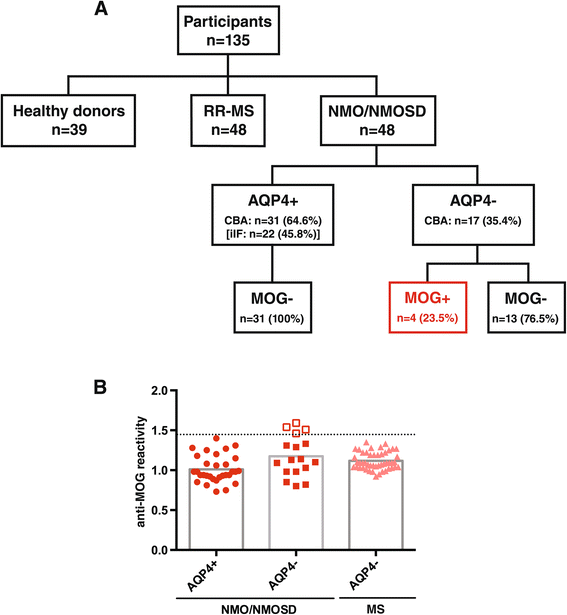
Even if assays with high specificity (≥99%) are used, true-positive (TP) results can easily be outnumbered by false-positive (FP) results if the prevalence of a marker is low and the number of samples tested is high. However, screening of large unselected populations for rare biomarkers generally decreases the positive predictive value of diagnostic tests by increasing the rate of false-positive results. Therefore, increasing numbers of patients with suspected or established MS are currently being screened for MOG-IgG. However, such misclassification has potential therapeutic implications: (a) similar to what has been observed in AQP4-IgG-positive NMOSD, some drugs approved for MS might be ineffective or even harmful in MOG-EM owing to differences in immunopathogenesis (b) MOG-EM is associated with a high risk of flare-ups after cessation of steroid treatment for acute attacks and may thus require close monitoring and careful steroid tapering and (c) patients positive for MOG-IgG might be particularly responsive to antibody-depleting treatments for acute attacks such as plasma exchange or immunoadsorption, to B cell-targeted long-term therapies such as rituximab, to treatment with intravenous immunoglobulins (IVIG) (especially in children ), and to immunosuppressive treatments. 
Accordingly, many patients with MOG-EM were falsely classified as having MS in the past. Importantly, however, MOG-EM and MS show a relevant phenotypic, i.e., clinical as well as radiological, overlap : like MS, MOG-EM follows a relapsing course in most cases, at least in adults, and 33 and 15% of adult patients with MOG-EM meet McDonald’s and Barkhof’s criteria for MS, respectively, at least once over the course of disease. īased on evidence from (a) immunological studies suggesting a direct pathogenic impact of MOG-IgG, (b) neuropathological studies demonstrating discrete histopathological features, (c) serological studies reporting a lack of aquaporin-4 (AQP4)-IgG in almost all MOG-IgG-positive patients, and (d) cohort studies suggesting differences in clinical and paraclinical presentation, treatment response and prognosis, MOG-IgG is now considered to denote a disease entity in its own right, distinct from classic MS and from AQP4-IgG-positive neuromyelitis optica spectrum disorders (NMOSD), which is now often referred to as MOG-IgG-associated encephalomyelitis (MOG-EM). While antibodies to MOG were originally thought to be involved in multiple sclerosis (MS), based on results from enzyme-linked immunosorbent assays employing linearized or denatured MOG peptides as antigen, more recent studies using new-generation cell-based assays have demonstrated a robust association of antibodies to full-length, conformationally intact human MOG protein with (mostly recurrent) optic neuritis (ON), myelitis and brainstem encephalitis, as well as with acute disseminated encephalomyelitis (ADEM)-like presentations, rather than with classic MS. Over the past few years, the role of immunoglobulin G serum antibodies to myelin oligodendrocyte glycoprotein (MOG-IgG) in patients with inflammatory CNS demyelination has been revisited. Finally, we provide recommendations regarding assay methodology, specimen sampling and data interpretation. In addition, we give a list of conditions atypical for MOG-EM (“red flags”) that should prompt physicians to challenge a positive MOG-IgG test result. In this paper, we propose indications for MOG-IgG testing based on expert consensus. To lessen the hazard of overdiagnosing MOG-EM, which may lead to inappropriate treatment, more selective criteria for MOG-IgG testing are urgently needed. However, screening of large unselected cohorts for rare biomarkers can significantly reduce the positive predictive value of a test. Accordingly, increasing numbers of patients with suspected or established MS are currently being tested for MOG-IgG. 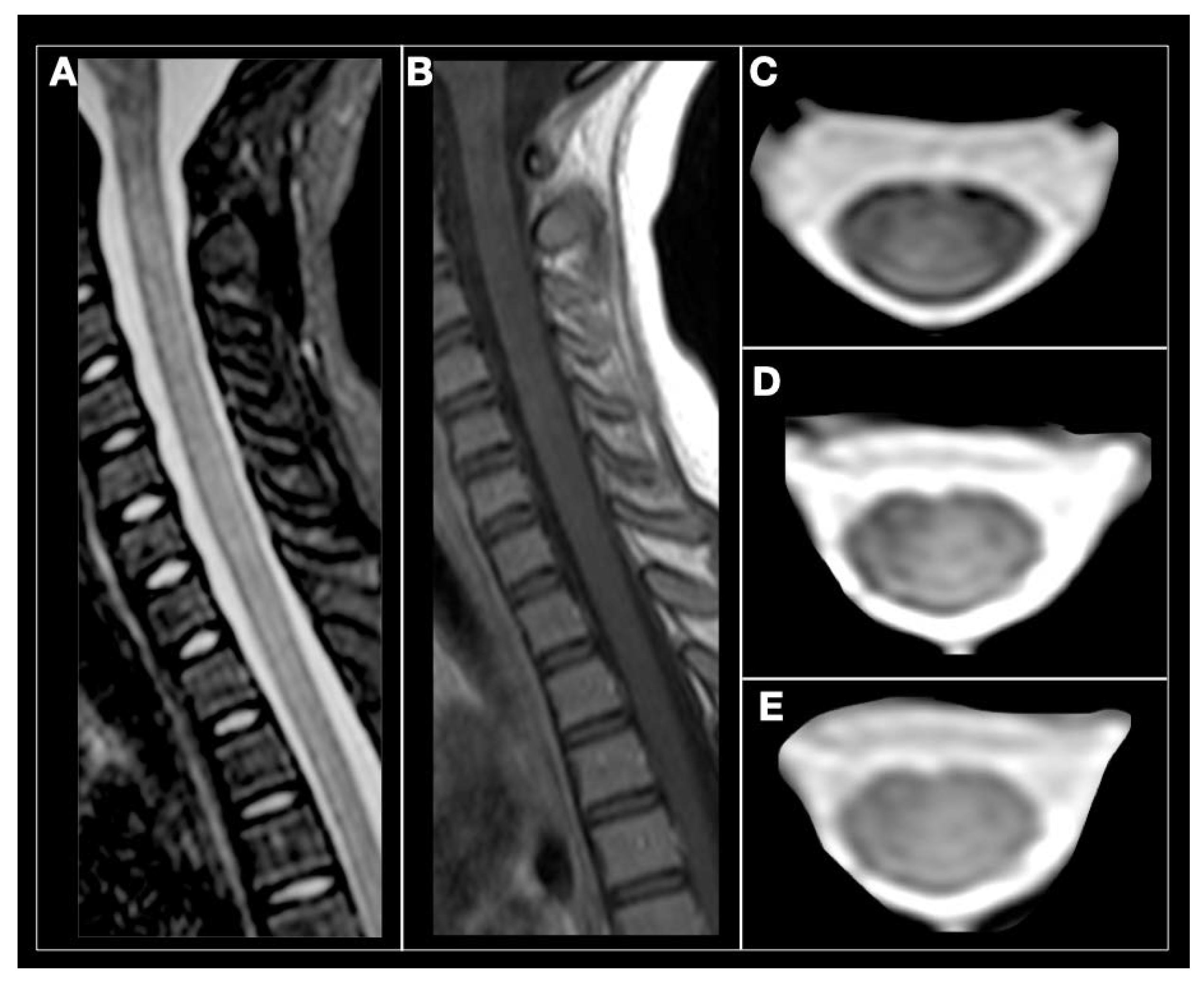
Owing to a substantial overlap in clinicoradiological presentation, MOG-EM was often unwittingly misdiagnosed as MS in the past. Most experts now consider MOG-IgG-associated encephalomyelitis (MOG-EM) a disease entity in its own right, immunopathogenetically distinct from both classic multiple sclerosis (MS) and aquaporin-4 (AQP4)-IgG-positive neuromyelitis optica spectrum disorders (NMOSD). Over the past few years, new-generation cell-based assays have demonstrated a robust association of autoantibodies to full-length human myelin oligodendrocyte glycoprotein (MOG-IgG) with (mostly recurrent) optic neuritis, myelitis and brainstem encephalitis, as well as with acute disseminated encephalomyelitis (ADEM)-like presentations. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
